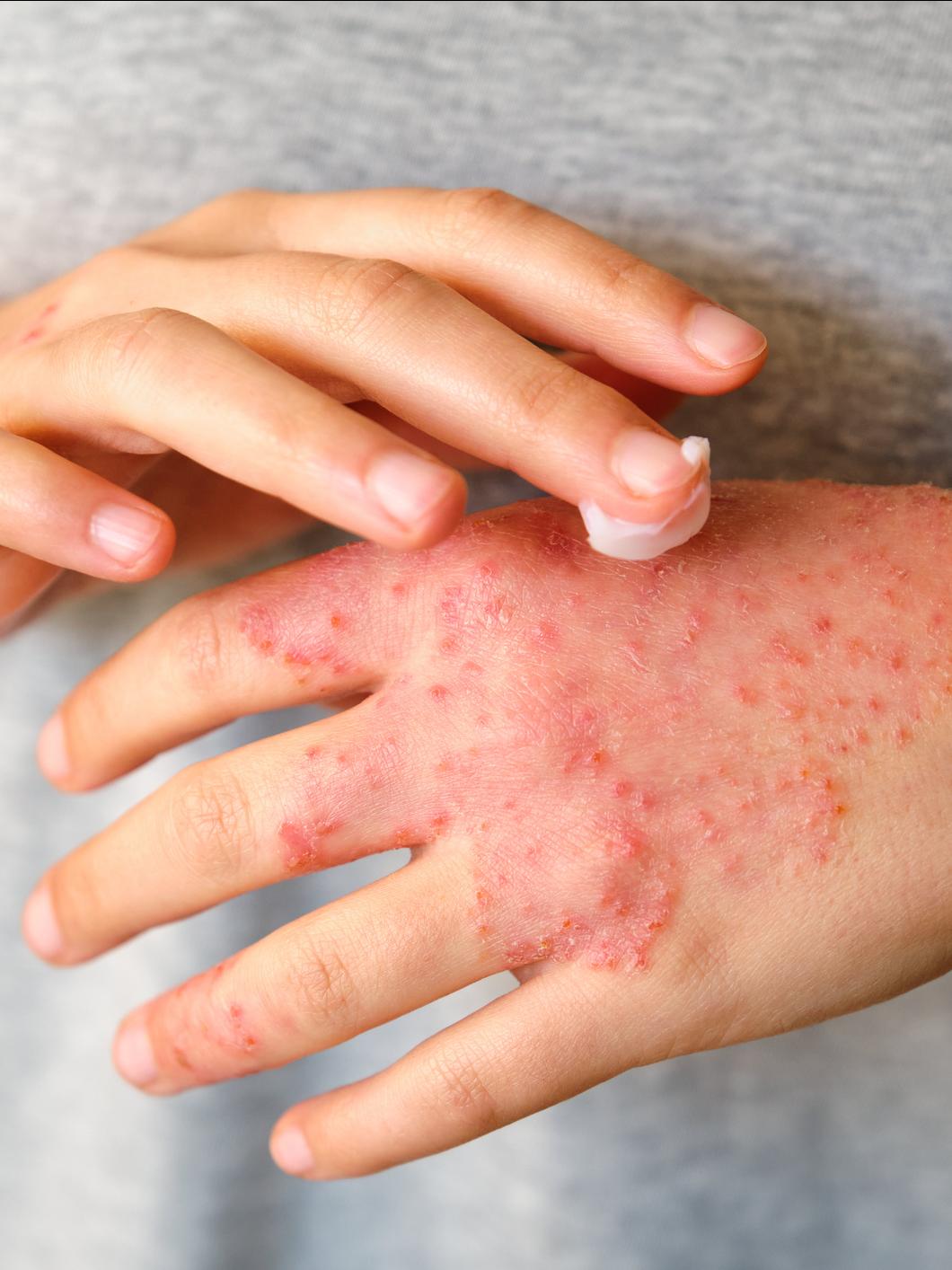
Add Your Voice
Community Voices in Research (CVR) is a community-powered registry that gathers information through surveys offering researchers a 360-degree view of what it is like to live with a bleeding disorder. Open to all persons affected by an inherited bleeding disorder as well as their non-affected family members (parents, spouses, grandparents, and siblings), it provides all the opportunity to participate in patient-reported outcomes research.
Connect with other families affected by bleeding disorders in your area through NBDF's chapter network.

“NBDF strengthens our community and gives us a sense that we belong to something bigger than ourselves—that we’re all in this together. That’s an amazing feeling.” — Monica, Community Member
Your generosity has a huge impact!